Water is everywhere in our daily lives, but few people think about how its density changes with temperature. This change is not just a science fact—it affects weather, ocean currents, ice formation, and even life on Earth. When you heat water or cool it down, the way its molecules move changes, and this causes the water to become heavier or lighter for its size. This simple idea creates big effects, from floating icebergs to the way your hot coffee cools down. Understanding how water density changes with temperature helps explain many natural phenomena, and it’s key for fields like climate science, engineering, and biology.
What Is Water Density?
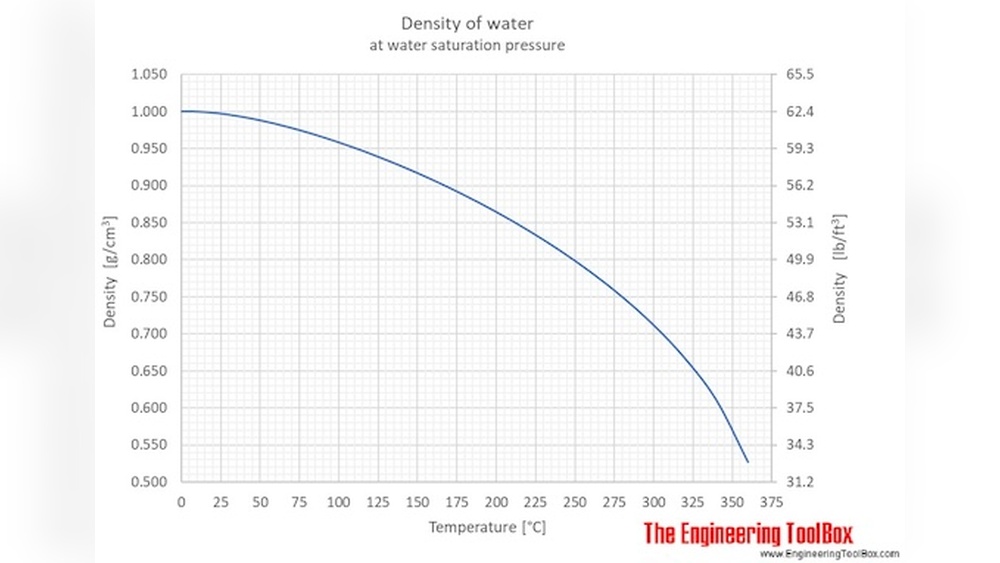
Density is how much mass is packed into a certain volume. For water, it means how heavy a certain amount of water is. Usually, scientists measure density in kilograms per cubic meter (kg/m³) or grams per cubic centimeter (g/cm³). Pure water at room temperature (about 20°C) has a density close to 1.0 g/cm³. But this number is not fixed; it changes as the water gets warmer or colder.
How Temperature Affects Water Density
When you heat water, the energy makes its molecules move faster. They start to push apart from each other, so the same amount of water now takes up more space. This means the density goes down. When you cool water, the molecules slow down, come closer together, and the density goes up.
However, water behaves a bit strangely compared to many other substances.
The Density Anomaly Of Water
Most materials get denser as they cool, but water is unique. When water cools down from, say, 25°C to 4°C, its density increases. But as you cool it further from 4°C to 0°C (its freezing point), its density actually decreases. That is why ice floats on water—solid water is less dense than liquid water.
This behavior is known as the density anomaly of water. It happens because of the way water molecules arrange themselves as they freeze, forming a crystal structure that takes up more space.
Water Density Values At Different Temperatures
To see how water density changes with temperature, look at the following values:
| Temperature (°C) | Density (g/cm³) |
|---|---|
| 0 | 0.99987 |
| 4 | 1.00000 |
| 10 | 0.99970 |
| 20 | 0.99820 |
| 40 | 0.99224 |
| 80 | 0.97180 |
| 100 (boiling) | 0.95835 |
Notice that the highest density is at 4°C. After that, as water gets warmer, the density keeps dropping.
Why Does Water Reach Maximum Density At 4°c?
The behavior of water at 4°C is not obvious. As water cools from higher temperatures, the molecules slow down and get closer, making the water denser. But below 4°C, hydrogen bonds force the molecules to start arranging themselves in a way that makes more open spaces—a preview of the structure that forms when water becomes ice.
So, the density actually drops as it gets colder than 4°C.
This property is very important in nature. For example, in lakes during winter, the coldest water (close to 0°C) floats on top and forms ice, while the denser water (at 4°C) stays below. This keeps aquatic life safe during freezing months because the water below the ice stays warmer.
Real-life Examples Of Water Density Changes
Oceans And Lakes
Ocean currents are driven in part by differences in water density, which are caused by both temperature and salt content. In lakes, as seasons change, the water layers mix because of density changes. This mixing brings nutrients up from the bottom, supporting fish and plants.
Weather And Climate
The way water density changes with temperature affects the formation of clouds, fog, and rain. When warm, less dense water rises and cooler, denser water sinks, it creates convection currents that help move heat around the planet.
Icebergs And Floating Ice
Ice floats because it is less dense than liquid water. This simple fact has a huge impact on Earth. If ice sank, ponds and lakes would freeze from the bottom up, killing aquatic life every winter. Instead, floating ice insulates the water below, allowing life to survive.
How Scientists Measure Water Density
Density is measured by finding the mass of a known volume of water. Scientists use special tools like hydrometers or pycnometers for precise measurements. For most practical purposes, a laboratory balance and a graduated cylinder are enough.
Let’s see a common method:
- Fill a graduated cylinder with a known volume of water (e.g., 100 mL).
- Weigh the cylinder with the water and subtract the weight of the empty cylinder.
- Divide the mass by the volume to get the density.
This process is repeated at different temperatures to see how density changes.
Water Density And Salinity
While temperature is a big factor, salinity (amount of dissolved salt) also affects water density. In the ocean, colder and saltier water is the densest and sinks to the bottom, driving global ocean currents.
Here’s a comparison:
| Water Type | Temperature (°C) | Density (g/cm³) |
|---|---|---|
| Freshwater | 4 | 1.00000 |
| Seawater | 4 | ~1.02500 |
This higher density in seawater is one reason why it’s easier to float in the ocean compared to a freshwater lake.
Importance Of Water Density In Engineering
Engineers must consider water density when designing ships, submarines, and water systems. For example, a ship must displace enough water to float, and this displacement depends on the water’s density. If a ship moves from cold water to warm water, or from freshwater to seawater, its buoyancy changes slightly.
This is why large ships adjust their loads based on the water they travel in.
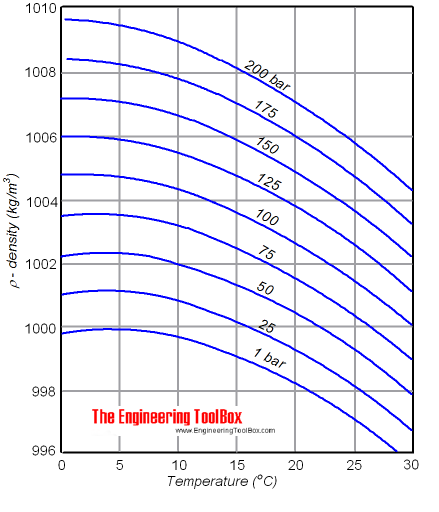
Credit: www.engineeringtoolbox.com
Water Density And Everyday Life
You may notice water density changes in small ways at home. For example:
- Ice cubes float in your drink because they are less dense than the liquid water.
- Hot water rises to the top in a pot, while cold water sinks.
- In swimming pools, the water at the bottom can feel colder because it is denser.
These examples show that water density isn’t just a science topic; it’s part of daily experiences.
How Water Density Affects Aquatic Life
Water’s density behavior protects aquatic life in cold climates. When a pond freezes, the ice forms on top and acts as an insulating layer. The densest water (at 4°C) stays at the bottom, giving fish and other organisms a safe place to survive the winter.
In the ocean, density-driven mixing brings nutrients from the deep to the surface, supporting the food web. Without these changes in density, many ecosystems would not exist as we know them.
.jpg)
Credit: www.geeksforgeeks.org
Water Density And Climate Change
As the climate warms, the temperature of lakes and oceans changes. This can shift the density layers, affecting how water mixes. If mixing patterns change, it can impact the availability of nutrients for marine life and alter weather patterns. Scientists track these changes closely to understand their effects on the planet’s health.
Water Density And Human Activities
Many industries rely on understanding water density:
- Power plants use water for cooling, and the way water absorbs and releases heat depends on its density.
- Brewing and food processing: Recipes and processes often depend on how water behaves at certain temperatures.
- Aquariums and fish farms: Keepers monitor water temperature and density to keep aquatic life healthy.

Credit: byjus.com
Common Misunderstandings About Water Density
Many people think water gets denser as it gets colder, all the way until it freezes. But, as explained, water’s maximum density is at 4°C, not 0°C. Another mistake is to ignore the effect of salt—seawater is always denser than freshwater at the same temperature.
A less obvious point: The density change with temperature is not linear. It slows down as you approach 4°C, then reverses as the temperature drops further.
Quick Reference: Water Density And Temperature
For fast comparison, here are the densities of pure water at several key temperatures:
| Temperature (°C) | Density (g/cm³) |
|---|---|
| 0 (just above freezing) | 0.99987 |
| 4 (maximum density) | 1.00000 |
| 25 (room temp) | 0.99705 |
| 50 | 0.98807 |
| 100 (boiling) | 0.95835 |
Notice the steady drop in density as temperature rises, but the peak at 4°C.
Non-obvious Insights About Water Density
Many beginners miss these points:
- Density below freezing: When water freezes, its density drops sharply. This is why icebergs float with most of their mass below the waterline.
- Thermocline formation: In deep lakes, rapid temperature changes with depth create a “thermocline,” a layer where the density changes quickly. This layer can trap nutrients and affect where fish live.
Understanding these details can help you predict how water will behave in real-world situations.
Frequently Asked Questions
Why Does Ice Float On Water?
Ice is less dense than liquid water. When water freezes, its molecules form a crystal structure that takes up more space, making ice lighter than the liquid it sits on. This is why icebergs float in the ocean.
At What Temperature Is Water Densest?
Water reaches its maximum density at 4°C (39.2°F). Above or below this temperature, its density drops, which is unusual compared to most liquids.
How Does Salt Affect Water Density?
Adding salt to water increases its density. That’s why seawater is denser than freshwater. The more salt dissolved, the heavier the water is for the same volume.
Why Is Water’s Density Important For Climate?
Water density changes drive ocean currents, which move heat around the globe. These currents affect weather patterns, climate, and the distribution of nutrients in the ocean.
Can Water Be Compressed To Change Its Density?
Water is nearly incompressible under normal conditions. Only at very high pressures—like deep in the ocean—does its density increase slightly. For most practical uses, temperature (and salinity) are the main factors changing water’s density.
Understanding how water density changes with temperature helps us explain everything from floating ice to global climate. This simple property is the foundation for many natural processes, and it’s a key concept in science and engineering. For a deeper look at water’s unique properties, visit the Wikipedia page on water properties. Next time you see ice floating in your drink or feel the cool water at the bottom of a lake, you’ll know the science behind it.

Hasan Al Sarker is a Reverse Osmosis Specialist. He has worked for many years to ensure safe drinking water for all. His research paper has been published in several journals, including Issue, Medium, and Slideshare. He is recognized as a water doctor among specialists though he did not attend medical college.
Besides working as a researcher of reverse osmosis technology, he is also very fancy with the kitchen and cooking. His guides are reading thousands of people every day. As a head of content, he is responsible for all the published articles at RO System Reviews.