Water is one of the most familiar substances in our lives, but it holds many secrets that surprise even scientists. One of the most interesting facts is that water expands when frozen. If you have ever left a bottle of water in the freezer, you may have seen it burst open or the ice pop out of an ice tray. This is not just a fun science trick—it’s a rare property that affects our planet in many important ways. Let’s explore why water behaves this way, what happens on a molecular level, and how this simple fact shapes everything from weather to living creatures.
The Basics: What Happens When Water Freezes
Normally, when things get cold, they shrink. Most liquids turn into solids by packing their molecules closer together. This makes them more dense. But water is different. When water cools down and reaches 0°C (32°F), it starts to freeze. As it freezes, its volume increases by about 9%. This means ice takes up more space than the water it came from.
This is why ice floats on water. When ice forms, it becomes less dense than liquid water. Density means how much mass fits into a certain space. Since ice is less dense, it stays on top of the water. This is a simple fact, but it has huge effects on nature and life.
The Science: Why Does Water Expand?
The Structure Of Water Molecules
To understand why water expands, we need to look at its molecules. A water molecule is made of two hydrogen atoms and one oxygen atom (H2O). These atoms are not in a straight line. They form a V-shape, with the oxygen in the middle and the hydrogens at an angle.
This shape allows water molecules to form hydrogen bonds with each other. A hydrogen bond is a weak link that connects one molecule to another. These bonds are not like strong chemical bonds inside molecules, but they are strong enough to affect how water behaves.
What Happens During Freezing
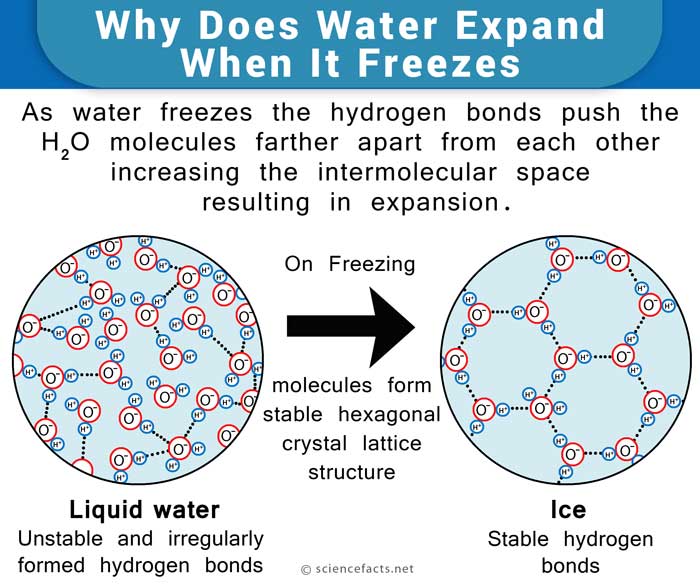
When water is liquid, the molecules move around and slide past each other. The hydrogen bonds form and break quickly, so the molecules can get close together.
When water freezes, the molecules slow down. The hydrogen bonds lock the molecules into a specific pattern. This pattern is called a crystal lattice. In ice, each water molecule connects to four others in a shape like a ring or cage. This structure leaves open spaces between molecules, making ice less dense.
Here’s a simple way to imagine it: Think of water molecules as people holding hands. In liquid, they move around and sometimes let go, so they can get close together. In ice, everyone holds hands in a wide circle, leaving more space between people. The circle takes up more space than a group standing tightly.
Data Table: Water Vs. Other Substances
How unusual is water’s behavior? Let’s compare water with some other common liquids.
| Substance | Volume Change When Frozen (%) | Solid State Density | Does Solid Float? |
|---|---|---|---|
| Water | +9% | 0.92 g/cm³ | Yes |
| Ethanol | -8% | 0.94 g/cm³ | No |
| Mercury | -3.7% | 13.69 g/cm³ | No |
| Benzene | -5% | 0.89 g/cm³ | No |
As you can see, most substances shrink and become denser when they freeze. Water is a rare exception.
Real-life Effects Of Water’s Expansion
Ice Floats: Why It Matters
Because ice is less dense, it floats on the surface of lakes, rivers, and oceans. This has big effects:
- Insulation for Life: In winter, ice forms a layer on top of water. This layer acts as insulation, keeping the water below from freezing solid. Fish and plants can survive under the ice, even in very cold places.
- Climate Regulation: Floating ice, like in the Arctic, helps reflect sunlight back into space. This keeps the planet cooler.
- Seasonal Changes: As ice melts in spring, it releases fresh water slowly, which is important for rivers and lakes.
Ice Damage: Cracks And Potholes
When water gets into small cracks in rocks and freezes, it expands. This process is called frost wedging. Over time, the expanding ice makes the cracks bigger. This is one reason why rocks break apart and why you see potholes in roads after winter.
Burst Pipes And Containers
If you leave a water bottle in the freezer, you might find it split open. The same thing happens to pipes in cold weather. As water inside freezes and expands, it can burst metal or plastic pipes. This can cause floods and expensive repairs.
Why Water Expansion Is So Unusual
Most materials contract when cooled. The fact that water expands is due to the special shape of its molecules and the way they link together. Only a few other substances, like silicon and bismuth, show similar behavior, but not as strongly as water.
Table: Substances That Expand When Frozen
| Substance | Expansion (%) | Common Uses |
|---|---|---|
| Water | +9% | Drinking, cleaning, agriculture |
| Bismuth | +3.3% | Alloys, fire detectors |
| Silicon | +2.6% | Electronics, glass |
Water’s expansion is much larger, and its effects are much more important for the environment.
Credit: www.sciencefacts.net
How Expansion Affects Weather And Nature
Lakes And Oceans
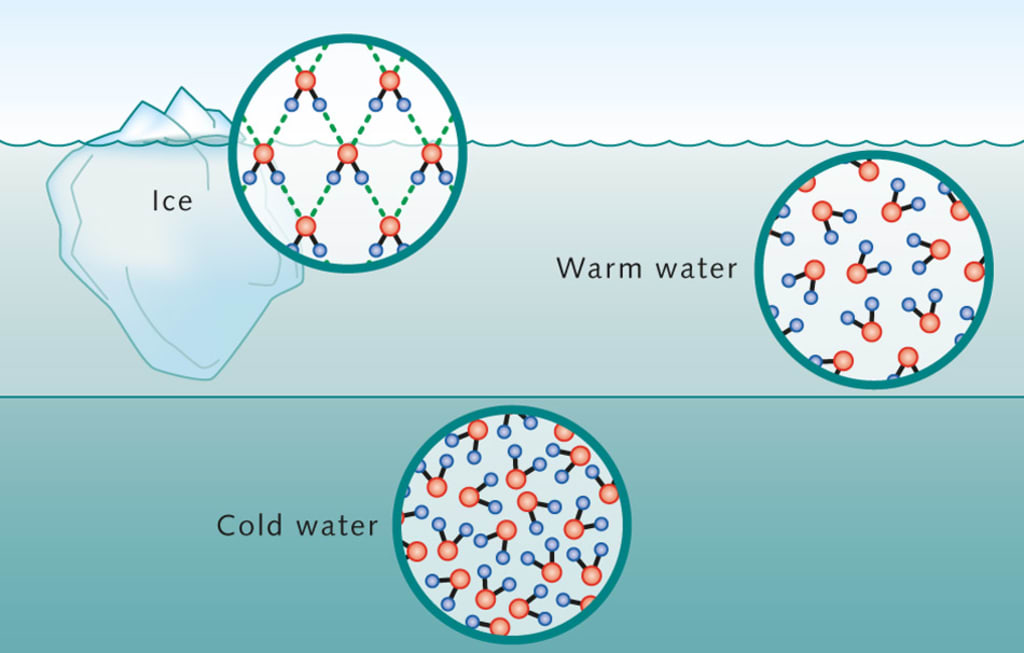
During fall, the surface water of lakes gets colder and sinks. But when it reaches 4°C (39°F), water is at its most dense. If it gets colder than this, it actually starts to rise, because it’s less dense. This means that in winter, the coldest water stays on top and freezes.
The water below stays warmer, which helps fish and plants survive.
Ice And Soil
In cold places, the ground can freeze. If there is water in the soil, it expands as it freezes. This can push up roads, rocks, and even buildings. This process is called frost heaving. Engineers must plan for this when building in cold climates.
Examples In Everyday Life
Freezer Mishaps
Anyone who has put a soda or water bottle in the freezer and forgotten it knows the result: a mess to clean up. This happens because as the water inside freezes, it needs more space. The bottle can’t stretch enough, so it splits.
Ice Trays
When you fill an ice tray, you may notice the ice cubes rise above the top. This is another sign of water’s expansion. The ice “grows” as it freezes.
Weathering Of Monuments
Over many years, water freezing inside cracks of statues and buildings can slowly break them apart. This is a big problem for old monuments and stone structures.
Two Non-obvious Insights
- Liquid Water Is Densest at 4°C: Most people think water gets denser as it cools, but water is actually heaviest at 4°C. Colder water starts to expand before it freezes, which is unusual. This is why deep lakes don’t freeze solid from the bottom up.
- Expansion Is Crucial for Mixing Nutrients: In spring, when ice melts, the “turnover” in lakes mixes oxygen and nutrients. This is important for fish and plant life. Without water’s special expansion, lakes would stay separated and become lifeless.
The Role Of Water Expansion In Human History
Agriculture And Soil
Water’s ability to break rocks helps create soil over thousands of years. As ice splits rocks, it makes the small pieces that plants need to grow. Without this slow process, there would be less soil, and farming would be harder.
Engineering Challenges
Engineers must design buildings, roads, and water systems to handle freezing water. They use special materials and designs to avoid damage. For example, water pipes in cold places are buried deep or wrapped to keep them from freezing.
Ice And Navigation
In cold climates, floating ice can block rivers and sea routes. This affects trade and travel. Icebreakers—special ships—are used to keep paths open.
How Scientists Study Water’s Expansion
Scientists use special tools to measure how much water expands when it freezes. They look at the structure of ice with microscopes and lasers. They also use computer models to predict how ice will behave in different conditions.
For more technical details, you can explore the research at Wikipedia’s Water Data Page.
Practical Tips: Avoiding Problems With Freezing Water
- Don’t Fill Bottles to the Top: Leave some space for expansion if you freeze water bottles.
- Insulate Pipes: In cold places, wrap pipes to keep them from freezing and bursting.
- Check Outdoor Faucets: Drain water from outdoor taps before winter to prevent damage.
- Be Careful with Glass: Never freeze water in glass containers—they are likely to break.
- Watch the Weather: In winter, potholes often appear after a freeze-thaw cycle. Drive carefully!
Water Expansion And Life On Earth
Without water’s expansion, life would be very different. Ice would sink, lakes and oceans could freeze solid, and many living things could not survive cold winters. The floating ice acts as a “blanket” for aquatic life and helps balance temperatures on Earth.
Frequently Asked Questions
What Causes Water To Expand When It Freezes?
Water expands when it freezes because of the way its molecules arrange themselves. In ice, water molecules form a crystal structure with open spaces. This structure takes up more space than the liquid form, making ice expand and float.
Why Does Ice Float On Water?
Ice floats because it is less dense than liquid water. As water freezes, it expands, so the same amount of molecules take up more space. This makes ice lighter per unit volume, so it floats.

Credit: chemistry.stackexchange.com
How Much Does Water Expand When It Freezes?
Water expands by about 9% when it freezes. This means that if you freeze one liter of water, the ice will take up about 1. 09 liters of space.
What Are Some Problems Caused By Water Expanding When Frozen?
When water expands as it freezes, it can burst pipes, crack rocks, and damage roads. This is why you see potholes in winter and why you need to winterize your plumbing in cold climates.
Credit: vocal.media
Is There Any Other Liquid That Expands Like Water When Frozen?
A few substances, like bismuth and silicon, also expand when frozen, but not as much as water. Water’s expansion is unique and has a bigger effect on life and the environment.
Water’s behavior when it freezes may seem simple, but it is at the heart of many natural and human processes. From protecting lakes to shaping landscapes, this small fact has a huge impact. Understanding why water expands when frozen helps us respect the hidden wonders of our everyday world.

Hasan Al Sarker is a Reverse Osmosis Specialist. He has worked for many years to ensure safe drinking water for all. His research paper has been published in several journals, including Issue, Medium, and Slideshare. He is recognized as a water doctor among specialists though he did not attend medical college.
Besides working as a researcher of reverse osmosis technology, he is also very fancy with the kitchen and cooking. His guides are reading thousands of people every day. As a head of content, he is responsible for all the published articles at RO System Reviews.

