When you look at a pond or a lake during winter, you may notice something interesting. The surface turns to ice first, while the water below stays liquid for a long time. Many people wonder why water freezes from the top down instead of from the bottom up or all at once. This question touches on physics, chemistry, and even biology. Understanding this process helps explain how life survives in cold climates and why frozen lakes behave differently from other liquids.
Let’s explore the science behind this special property of water, how it works, why it matters for nature and daily life, and practical tips to observe and manage freezing water. As someone who has studied and taught environmental science for over a decade, I will also share real-world examples, key measurements, and practical insights to make these ideas clear.
The Science Behind Water Freezing From The Top
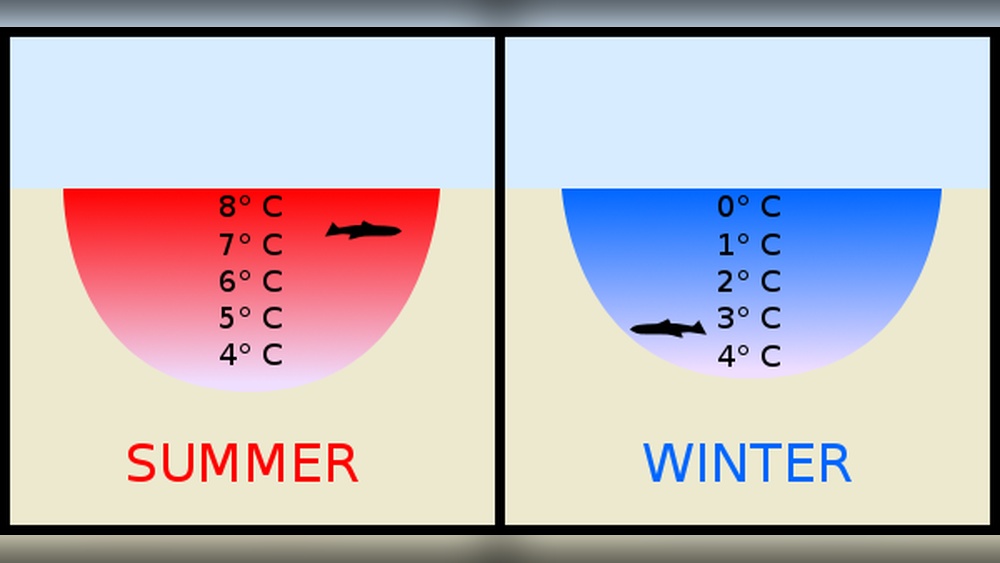
Water is not like most other liquids. Most substances become denser and heavier as they cool, so they freeze from the bottom up. Water, however, has a special property called the density anomaly.
When water cools, it becomes denser until it reaches about 4°C (39.2°F). Below this temperature, water actually becomes less dense as it approaches freezing point (0°C or 32°F). This is because water molecules arrange themselves in a way that creates more space between them as they form a solid structure (ice). As a result, ice floats on liquid water.
This unique behavior is due to hydrogen bonding. Each water molecule can form four hydrogen bonds with neighboring molecules, creating a lattice structure when it freezes. This lattice is open and less compact than liquid water, so it floats.
How The Freezing Process Works
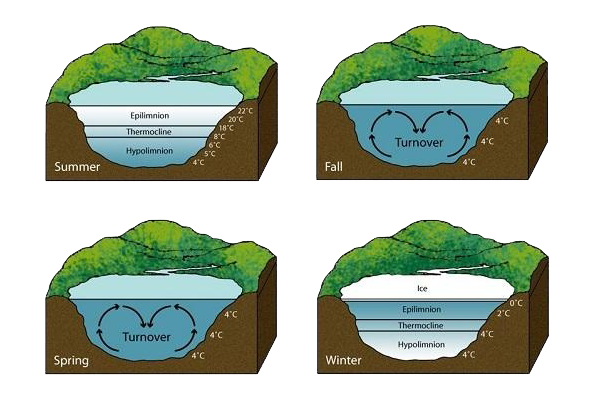
The freezing of a lake or any large body of water follows a step-by-step pattern, mainly driven by temperature and density.
- Cooling at the Surface: As air temperature drops, the water at the surface cools first.
- Sinking of Denser Water: When the surface water cools to 4°C, it becomes denser and sinks, pushing slightly warmer water up.
- Formation of Ice: Once all water at the surface is at 4°C, further cooling causes the surface water to drop below 4°C. Now, instead of sinking, this colder water stays at the top and eventually reaches freezing point.
- Ice Layer Grows Downwards: Ice forms at the surface and thickens downward as the cold air continues to remove heat.
Here is a summary of the main steps:
| Step | Temperature Range (°C) | What Happens |
|---|---|---|
| Surface Cooling | Above 4°C | Surface cools, denser water sinks |
| Maximum Density | 4°C | Coldest, densest water at bottom |
| Freezing Begins | 0°C | Surface water freezes, ice forms at top |
| Ice Thickens | Below 0°C (air) | Ice layer grows from top down |
Why This Matters In Nature And Daily Life
The way water freezes is not just a scientific curiosity. It has real consequences for ecosystems, safety, and engineering.
- Life in Water: If lakes froze from the bottom, fish and other life would have nowhere to survive during winter. The top ice acts as an insulating blanket, keeping water below at a stable temperature.
- Ice Safety: The strength and thickness of surface ice are important for activities like skating, fishing, and walking on lakes.
- Infrastructure: Pipes, dams, and water tanks are designed with this property in mind to prevent damage from expanding ice.
Practical Examples And Real-world Data
Let’s look at real measurements and events that show how this process works.
- In Lake Superior (USA), surface ice can reach 0.5 to 1 meter thick in winter, while the bottom water remains at about 4°C.
- During the 2014 cold wave, many North American lakes froze at the top but still supported fish and aquatic life beneath the ice, as confirmed by temperature sensors.
- In home freezers, water in a cup will start to freeze at the top and form a layer before the rest of the water turns solid.
These examples highlight the importance of the top-down freezing process and how it protects life and structures.
Credit: www.ausableriver.org
Key Signs And Indicators Of Top-down Freezing
Recognizing when water is freezing from the top can help you predict ice safety and quality.
- Clear Ice Layer: The first sign is a thin, clear ice sheet on the surface.
- Bubbles Trapped: Air bubbles may be trapped under the ice, showing the freezing direction.
- Temperature Drop: Surface temperature drops before the rest of the water cools.
- No Ice at Depth: Deeper water remains liquid even when the surface is frozen.
If you monitor a pond or even a cup of water outside, you can observe these indicators as the freezing progresses.
Step-by-step Guidance To Observe And Test Freezing
If you want to see this process yourself, here is a simple way to do it.
- Fill a transparent container with tap water and place it outside on a cold night.
- Measure air and water temperature with a simple thermometer.
- Check every hour to see where ice forms first.
- Note the thickness of the ice at the top and whether the bottom remains liquid.
- Record changes as the freezing continues. The surface should freeze long before the bottom.
For a more advanced test, use two thermometers, one near the surface and one at the bottom, to compare temperatures as ice forms.

Credit: www.youtube.com
Common Problems And Solutions
People sometimes misunderstand or face issues related to water freezing. Here are a few common problems and how to address them:
Problem: Pipes bursting in winter.
- Solution: Insulate pipes and allow a slow drip to keep water moving, since ice forms at the coldest exposed spots first.
Problem: Unsafe ice on lakes.
- Solution: Wait for at least 10 cm (4 inches) of clear, solid ice before walking or skating. Measure ice thickness at several points.
Problem: Fish kills in frozen ponds.
- Solution: Maintain a hole in the ice to allow gas exchange. A floating heater or bubbler can help.
Problem: Cracks in ice.
- Solution: Cracks are normal as ice expands and contracts. Avoid areas with visible cracks for safety.
Maintenance Tips For Frozen Water Bodies
To keep lakes, ponds, or even home water features safe and healthy during winter, follow these maintenance tips.
- Monitor ice thickness regularly with an auger or drill.
- Clear snow off the ice to allow sunlight to reach the water and promote oxygen for fish.
- Keep a small area open for gas exchange if you have fish or plants.
- Check for leaks in pipes and water tanks before freezing weather starts.
- Store water containers indoors if possible to prevent cracking.
These steps help you manage freezing water safely and effectively.

Credit: www.seacoastonline.com
Key Data Table For Water Freezing
Here is a quick reference with measurable values related to water freezing:
| Parameter | Value | Notes |
|---|---|---|
| Maximum Density Temp | 4°C (39.2°F) | Heaviest water sinks |
| Freezing Point | 0°C (32°F) | Ice forms at surface |
| Safe Ice Thickness (walking) | 10 cm (4 in) | Clear, solid ice only |
| Common Ice Thickness (lakes) | 0.5–1 m (20–40 in) | Varies by region |
| Hydrogen Bonds per Molecule | 4 | Forms open lattice in ice |
Frequently Asked Questions
Why Does Ice Float On Water?
Ice floats because it is less dense than liquid water. The molecules in ice are arranged in a way that takes up more space, making it lighter than the same volume of liquid water.
Can Lakes Freeze Solid All The Way To The Bottom?
In most climates, lakes do not freeze to the bottom. The ice at the top insulates the water below, keeping it near 4°C. Only very shallow lakes in extremely cold regions might freeze solid.
How Thick Must Ice Be To Walk On Safely?
For safe walking, ice should be at least 10 centimeters (4 inches) thick. Always check thickness in several spots and avoid discolored or cracked ice.
Why Is The Freezing Point Of Water 0°c?
Water freezes at 0°C under normal atmospheric pressure. This is due to the energy required for water molecules to form stable hydrogen-bonded ice crystals.
Does Salt Affect How Water Freezes?
Yes, adding salt lowers the freezing point of water. This is why salt is used on roads to prevent ice formation and why ocean water freezes at about -1.8°C (28.8°F).
Key Takeaway
Water’s unique behavior—freezing from the top down—protects aquatic life, shapes our winter environment, and influences engineering decisions. By understanding the science, recognizing the signs, and following safety and maintenance tips, you can manage and enjoy frozen water bodies with confidence.
For further reading and detailed scientific explanation, see this Wikipedia article on water and the USGS Water Science School.

Hasan Al Sarker is a Reverse Osmosis Specialist. He has worked for many years to ensure safe drinking water for all. His research paper has been published in several journals, including Issue, Medium, and Slideshare. He is recognized as a water doctor among specialists though he did not attend medical college.
Besides working as a researcher of reverse osmosis technology, he is also very fancy with the kitchen and cooking. His guides are reading thousands of people every day. As a head of content, he is responsible for all the published articles at RO System Reviews.